
When these two conditions are met, the equation is said to be balanced. Also the sum of the charges on one side of the equation must be equal to the sum of the charges on the other side. This means that a chemical equation must have the same number of atoms of each element on both side of the equation. The Law of Conservation of Mass states that mass is neither created nor destroyed in an ordinary chemical reaction. If the elements in a chemical formula are properly capitalized, the smart case converter leaves them as you have typed.Ī balanced chemical equation accurately describes the quantities of reactants and products in chemical reactions. The equation can be written in lowercase letters.To enter the equation sign, you can use either "=" or "->" or "→" symbols.To enter charge species, just type them as they are, for example Hg2+, Hg22+, or Hg2^2+.(ii) Sodium hydroxide solution(in water) reacts with hydrochloric acid solution(in water) to produce sodium chloride solution and water. (i) Solutions of barium chloride and sodium sulphate in water react to give insoluble barium sulphate and the solution of sodium chloride. Note that the state symbols are written in brackets after the chemical formula. All types of parentheses are correct, for example K3 Write a balanced chemical equation with state symbols for the following reactions. For example, for the reaction between sodium and water, this is the symbol equation with state symbols.Spaces are irrelevant, for example Cu SO 4 is equal CuSO4. 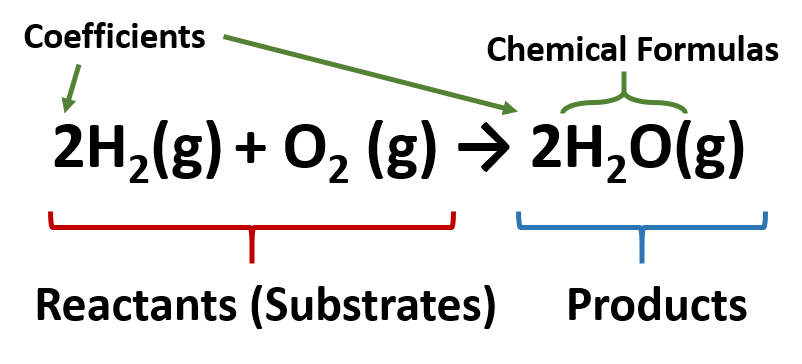
Remember that if there's no coefficient in front of an element, it's assumed that the coefficient is 1.Enter an equation of a chemical reaction and click 'Submit' (for example: so32-+cr2o72->cr3++so42- ). Now the number of atoms in each element is the same on both sides of the equation, so the equation is balanced. To balance this, add the coefficient 2 before H2 on the left side of the equation so there are 4 hydrogen atoms on each side, like 2H2 + O2 → 2H2O. However, subscripts can't be changed and are always multiplied by the coefficient, which means there are now 4 hydrogen atoms on the right side of the equation and only 2 hydrogen atoms on the left side. For the equation H2 + O2 → H2O, you would add the coefficient 2 before H2O on the right side so that there are 2 oxygen atoms on each side of the equation, like H2 + O2 → 2H2O. To balance the equation, you'll need to add coefficients to change the number of atoms on one side to match the other. Since the number of atoms in each element isn't identical on both sides, the equation is not balanced. There are 2 hydrogen atoms and 1 oxygen atom on the right, so you would write "H=2" and "O=1" under the right side. Calculator designed to balance chemical equations with results of: the balanced equation, word equation, and how it happened. For the equation H2 + O2 → H2O, there are 2 hydrogen atoms being added to 2 oxygen atoms on the left, so you would write "H=2" and "O=2" under the left side. For example, your equation should look something like "H2 + O2 → H2O." Count the number of atoms in each element on each side of the equation and list them under that side. To balance a chemical equation, first write out your given formula with the reactants on the left of the arrow and the products on the right. If the value you assigned returns coefficients that have a greatest common factor (GCF), simplify the chemical equation by dividing each value by the GCF.If there is only one fraction, multiply all values by that values denominator. If the value you assigned returns fractional values, just multiply all values by the least common multiple (LCM) of the denominators to get rid of the fractions. 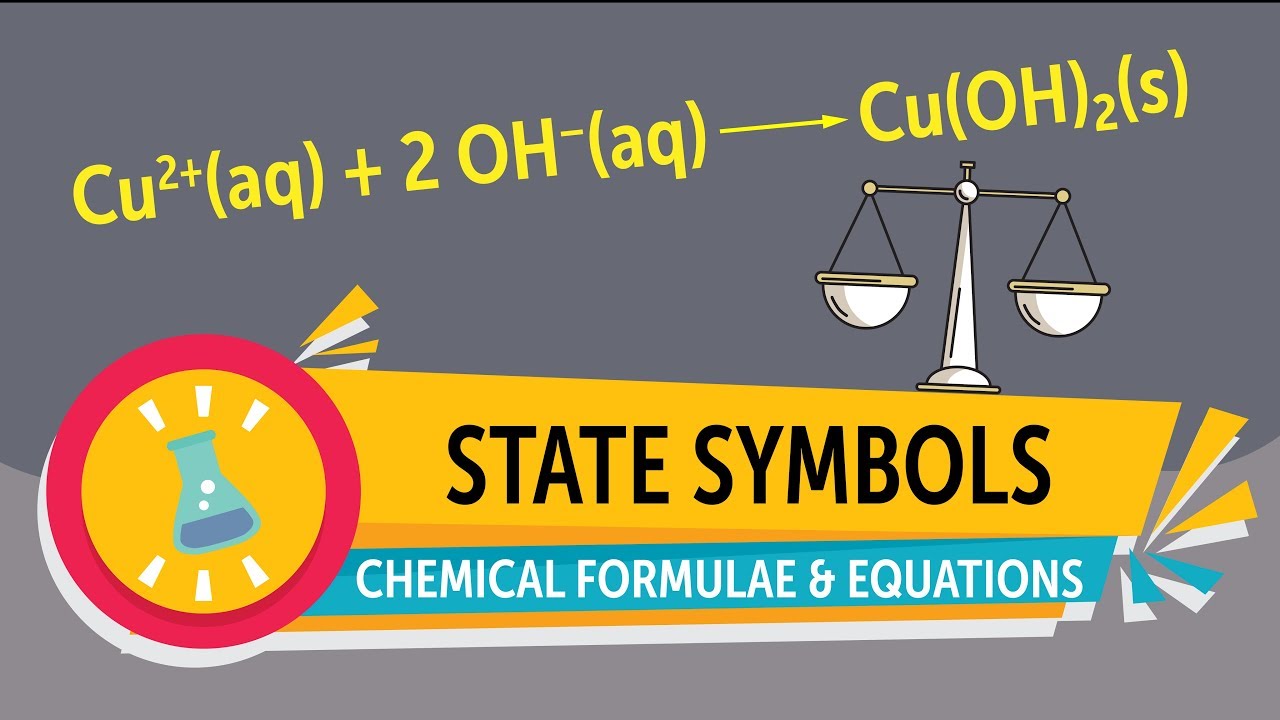
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
